 
Suppose $d$ represents the amount of Carbon $14$ in the plant at its time of death while $b$ represents the total amount of Carbon $12$. The inherent difficulties with mathematical modeling, some of which are mentioned in the previous paragraph as regards this particular example. If the teacher has the time and inclination, it also reveals many of Interesting and important example of mathematical modeling with an exponential function. This problem is intended for instructional purposes only. More recent technology actuallyĪllows scientists to measure the remaining Carbon $14$ much more accurately. This is done by measuring the current decaying Carbon $14$ then it is not statistically reliable with very small samples. The method used to estimate the amount of Carbon $14$ in a given sample. Means that extremely precise measurements will be needed to determine how much Carbon $14$ is in a specimen. This is a serious issue because the current ratio of $1$ to $1,000,000,000$ The accuracy of measurement for the amount of Carbon $14$ in a given sample. Such as volcanoes, and in recent times, by human activity. These levels can be influenced by climate, by natural processes 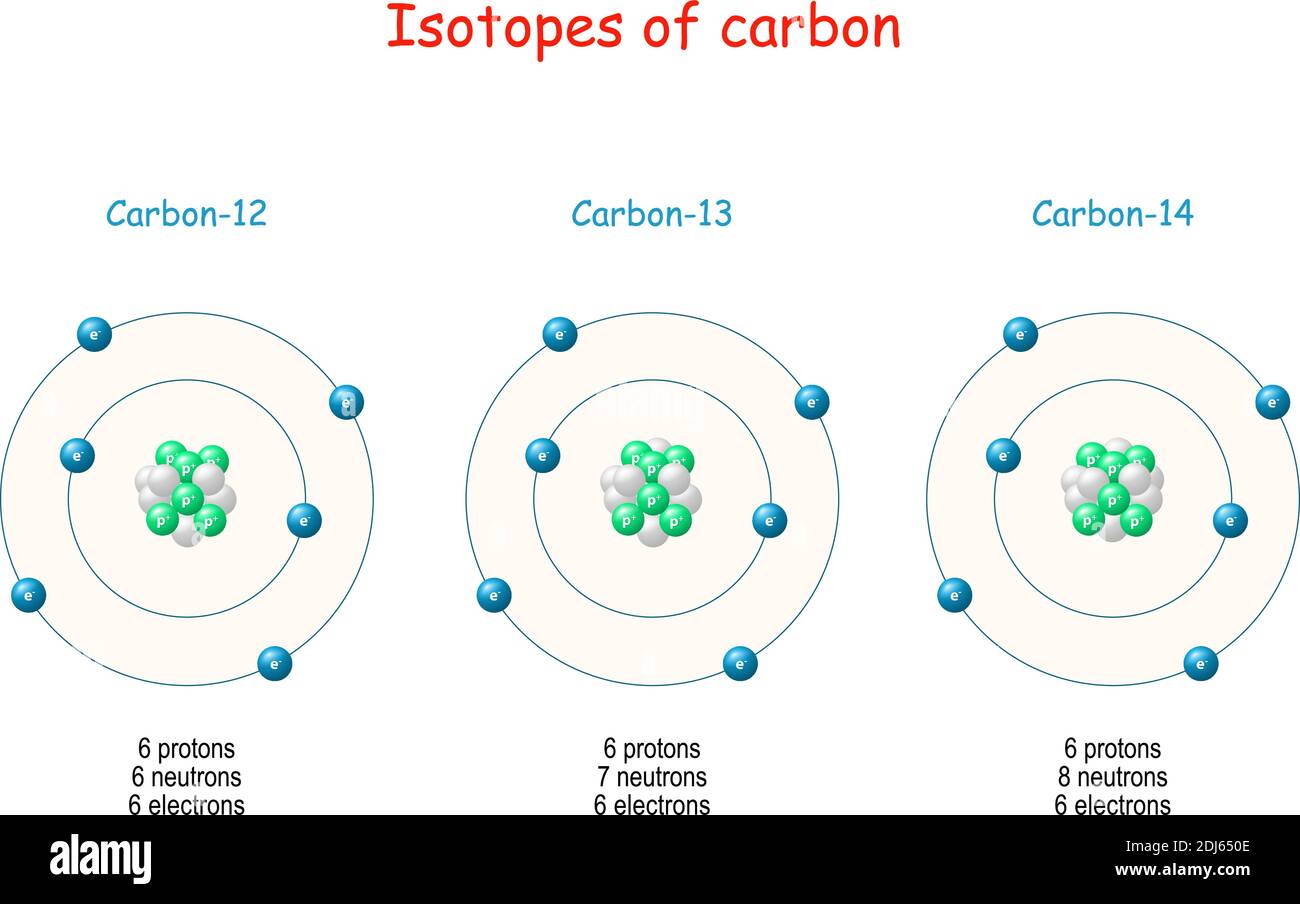
The hypothesis that levels of Carbon $14$ in the environment have been relatively constant. Many factors limit the accuracy of using Carbon 14 for dating including Organic material has ceased to absorb Carbon $14$, that is, as soon as it dies.Ĭarbon $14$ dating is a fascinating topic and much information can be This ratio decreases, hypothetically, at a constant exponential rate as soon as the In the sample but rather on the ratio of Carbon $14$ to Carbon $12$. It is based not on the amount of the Carbon $14$ isotope remaining This problem introduces the method used by scientists to date certain organic Using this information and the given half-life of Carbon 14, explain why this method of dating is not used for dinosaur remains. What can you concludeĭinosaurs are estimated to have lived from about $230,000,000$ years ago In a particular preserved plant, the ratio of Carbon $14$ to Carbon $12$ isĮstimated to be about $1$ to $13,000,000,000$. Write an equation for a function which models the ratio of Carbon $14$ to Carbon $12$ in a preserved plant $t$ years after plant has died. Scientists estimate that the ratio of Carbon $14$ to Carbon $12$ today is approximatelyĪssuming that this ratio has remained constant over time, The Carbon $14$ within the organism decays exponentially, becoming Nitrogen $14$, with a WhenĪn organism dies, it ceases to absorb Carbon $14$ from the atmosphere and Of Carbon $14$ to Carbon $12$ in the artifact or remains to be dated. In order to use Carbon $14$ for dating, scientists measure the ratio 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
